

ADP Diagnostics works to bring you the latest innovative and high quality products for your Laboratory, Health Centre and Doctor's Surgery. From Best In Class Point Of Care equipment to WORLD FIRST, CUSTOM MANUFACUTURE and high quality QC material.
If you cannot find a product you need for your laboratory, please contact us and we will strive to find it, or design and manufacture it for you.
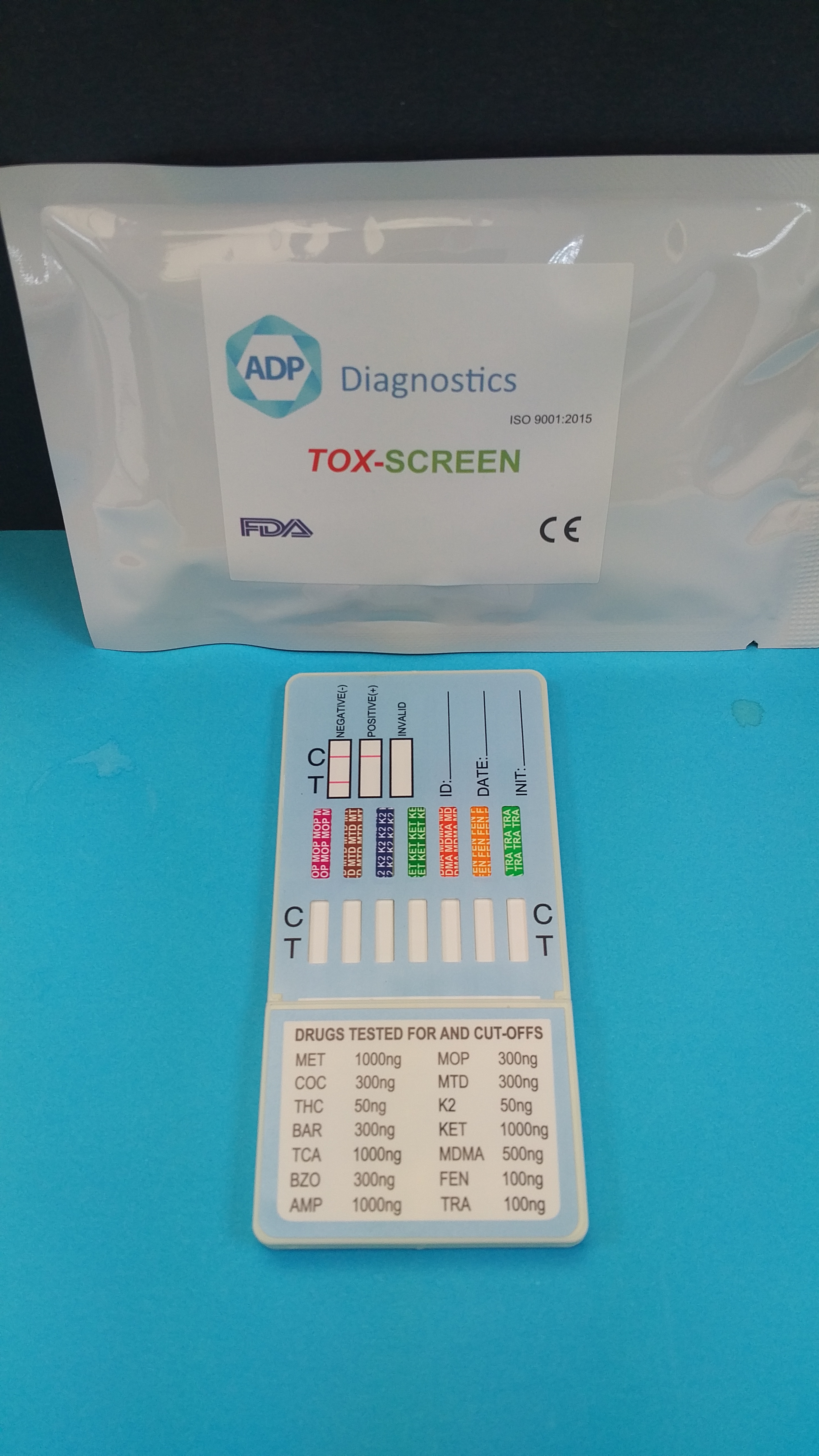
We are excited to introduce our new TOXSCREEN 16 Panel Rapid Test. Unique in Ireland an UK
After 5 years of our 12 Panel ToxScreen, introduced to cover the rising abuse of synthetic cannabis (K2 / SPICE), and Ketamine, we are excited to introduce an update to our unique 14 Panel ToxScreen, to a 16 Panel. New drugs added due to requests from our customers, Gabapentin and Pregabalin our 16 panel brings together some of the more "tradtionally" abused drugs with those which are more recent and emerging.
Drugs included are: MET / COC / THC / BAR / TCA / BZO / AMP / OPI / EDDP / K2 / KET / MDMA / FEN / TRA / GAB / PGB
Available to order now

A Tri- level control for the monitoring of AMH assays
Ensure accurate monitoring of patient samples for AMH by running this QC daily. The levels of our control are targeted to check the precision of the assay at the bottom of the measuring range, at the clinical cut-off and higher in the measuring range for linearity.
MORE INFORMATION:
TRI-LEVEL, 3 X 2 X 2 ML
LIQUID FROZEN, 12 MONTHS EXPIRY FROM MANUFACTURE
CAT NO: QC AMH

Haemaglobin A1C, Whole Blood Quality Control
A true whole blood HbA1C Bi-Level Quality Control. Manufacutred for optimum performance on HPLC systems such as BioRad and Menarinni. This control also works excellently on the Sebia A1C system and with immunochemistry assays.
Supplied as a two level control with 3 bottles of each level.
18 months expiry from manufacture.
Kit size 3 x 2 x 0.5mls
Catalogue Number, QC HBA

The ADP ‘PSA Ultra Low level’ Quality Control is designed to monitor the performance of PSA in immunoassays.
ADP’s ‘PSA Ultra Low level’ control is manufactured as stabilised serum from human origin. The concentration of the QC has been chosen to ensure optimal assay performance at a critical sensitivity level. Typical level, Roche Cobas, 0.065 ng/ml
Designed to aid in the monitoring patients, 'Post Prostatectomey' to ensure the potential ultra low levels reported by the immunoassays are correct. "A low level or undetectable PSA level is defined as ≤0.06 ng/mL" Reference: Int. J. Radiation Oncology Biol. Phys Vol.37 No.5 pp 1035-1041, 1997
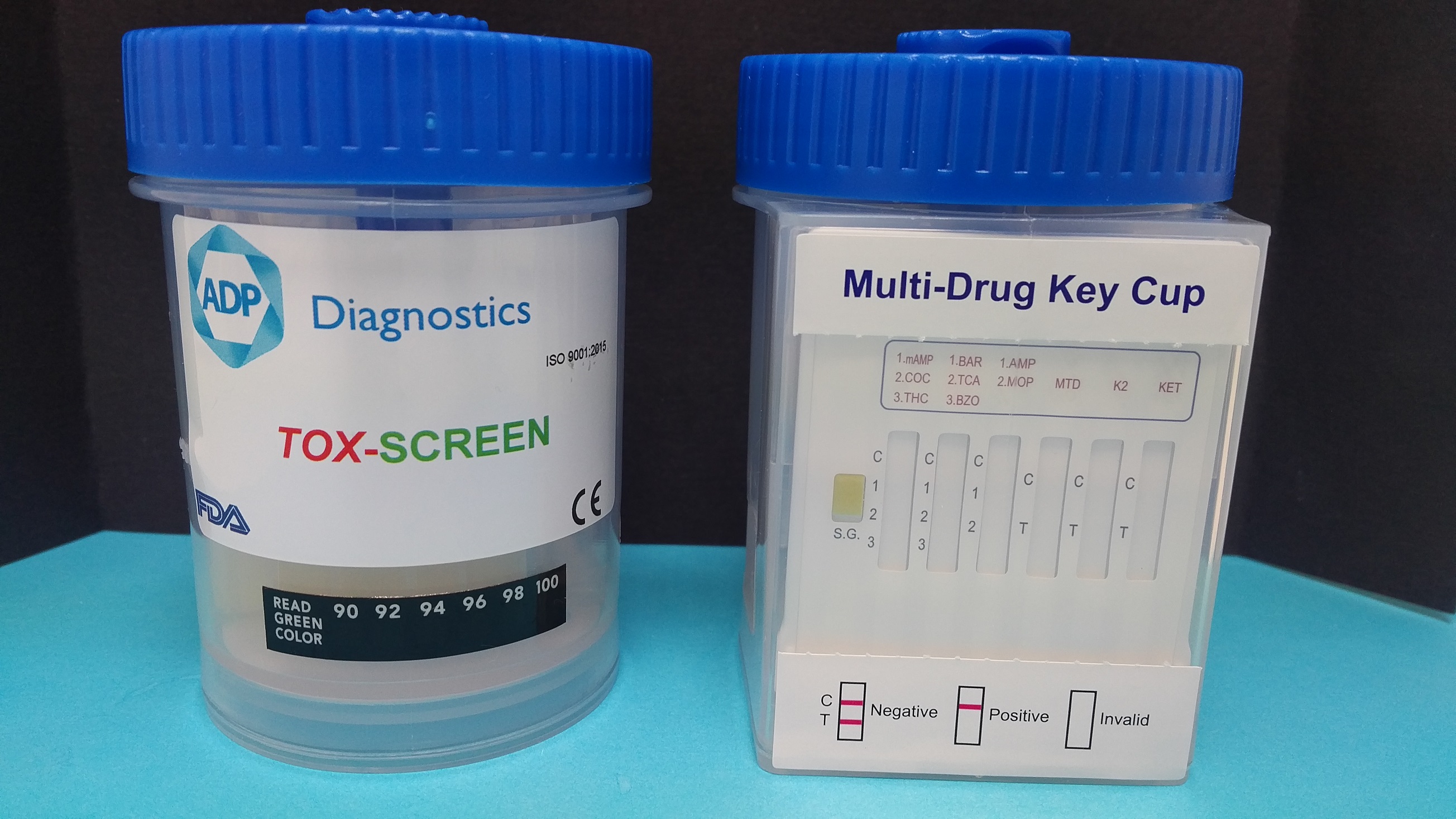
16 Parameter Secure Cup Drugs of Abuse Screening Test
An easy to use DOA screening test with results available in 5 minutes.
The design of this screening test covers all popular abused drugs, PLUS newer drugs such as K2/SPICE and Ketamine.
Also included is an adulterant check, utilising Specific Gravity.
Drugs in the 12 parameter screen are;
MET / COC / THC / BAR / TCA / BZO / AMP / OPI / EDDP / K2 / KET / MDMA / FEN / TRA / GAB / PGB
2 years expiry from manufacture
Supplied in pack of 25 or an Over the Counter Single and Twin Pack.

Manufactured in the U.S.A.
We have been selling these products for almost 10 years with great sucess.
Quality is paramount in the products we choose, and with over 20 years experience in selling Quality Controls, these are some of the best in the world.
Most Controls are liquid stable, with very long 'Open Vial' stability, typically 30 days, and expiry dates if 2 to 3 years.
Products include, Cardiac Controls, with a brand new product, dedicated to the Siemens Stratus CS analyser, Immunoassay, Tumour Markers, Immunology and Urine Controls.

The new DocUReader 2 Pro is a compact urine analyser designed for doctor’s offices and smaller laboratories to read and evaluate the LabStrip U11 Plus urine test strips. Although the instrument is intended for in vitro diagnostic use by qualified physicians and laboratory staff, no special training is required to use them.
The easy to use software features all the Point Of Care requirements of a modern laboratory, including Operator and QC lock-out, strip verification request, Patient I.D. and Operator I.D. scanning.
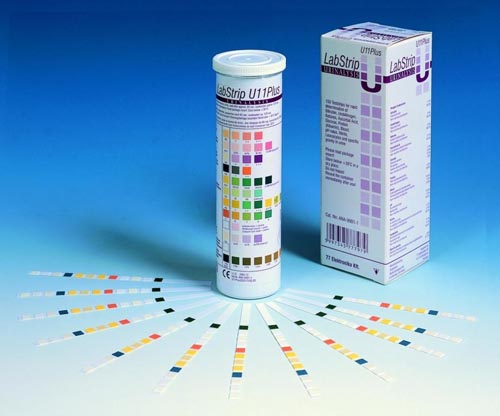
The LabStrip U11 Plus and LabStrip U11 Plus GL high-grade, multiparameter urinalysis test strips are in vitro diagnostic medical devices for professional laboratory use. They are designed for semi-quantitative urine analysis from fresh urine. LabStrip U11 Plus and LabStrip U11 Plus GL test strips are intended to be used in screening tests for liver disease, biliary and hepatic obstructions, diabetes, and haemolytic, urological, and nephrological diseases associated with hematuria or hemoglobinuria, diseases of the kidneys and urinary tract, and pathologic shifts in pH, as well as for the investigation of urinary sediment.


Possibility of qualitative tests measurements Available quantitative tests
AFP Cystatin C HS-Insulin Transferrin
CA-125 C-Peptide LH Troponin I
CA 15-3 D-Dimer Microalbumin TSH
CA 19-9 FABP Myoglobin HS-TSH
Calprotectin Ferritin NT-proBNP ULTRA-TSH
CEA FOB Procalcitonin
CK-MB FSH Prolactin Coming soon
Cortisol HbA1c HS-Prolactin Progesterone
COVID-19 spike IgM/IgG Hb-Total PSA (quantitative Testosterone
CRP (laboratory or doctor’s hCG (S/P or U) or semi-quantitative) Vitamin D office) beta-hCG PSA Free