

28 October 2016
Every day in hospital laboratories across the world, millions of patient samples are being scanned on automated analysers for compromised samples. The ‘flag’ rate for compromised samples from Serum Indices (Haemolysis, Icterus and Lipaemia) can be up to 10%. However, how do we know that the automated analysers are accurately scanning the samples for Serum Indices?
Until recently, there has been no way to routinely check the indices-scanning performance of automated analysers. As well as this, developmental trials have shown differences of up to 10% between ‘analytical lines’. One analyser can exclude 10% more results than another analyser even in the same laboratory.
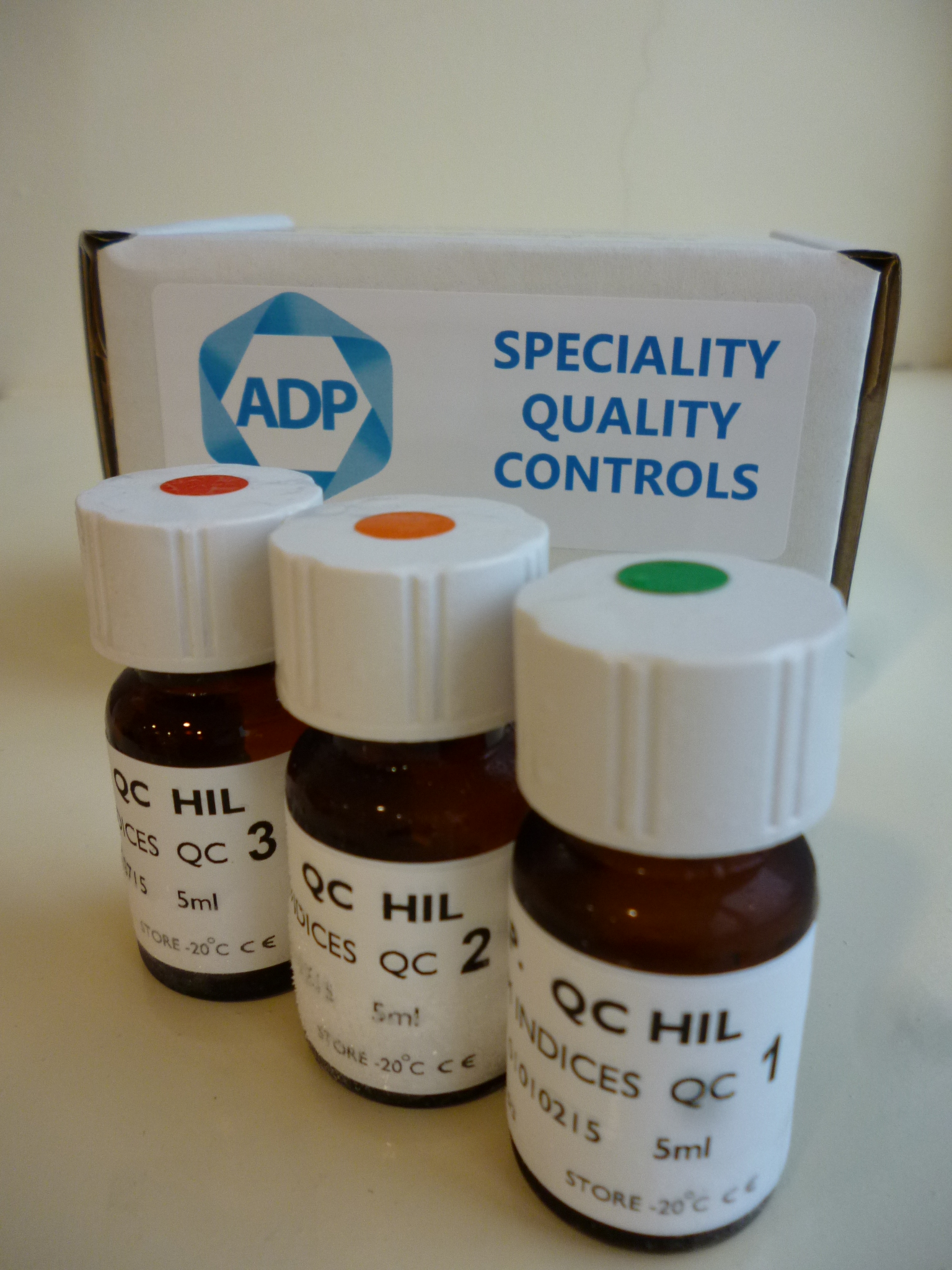
ADP Diagnostics has developed the world’s first Serum Indices Quality Control material to allow laboratories to check that their analysers are accurately scanning and flagging compromised samples.
The Serum Indices Quality Control is available as a frozen liquid stable product with all 3 parameters combined into one control. The control is available in 3 levels, 1+, 2+ and 3+. We have developed instrument specific controls suitable for Roche, Abbott and Beckman analysers. Pack size is 4 x 3 levels x 5ml.
Our Serum Indices Quality Control is the only 3rd party QC sera available on the market and is therefore essential for any laboratory that needs to comply with national and international standards, such as ISO 15189 accreditation.
The product is currently being used by hospital laboratories throughout the UK and Ireland, Europe and Australia.
ADP Diagnostics will have the official international launch of the Serum Indices Quality Control sera at Medica 2016 in November where we will be participating as an exhibitor. For more information on the product and market feedback, please visit our stand in Hall 1 F07.

12 April 2016
ADP are proud to announce the release of our 3rd party Quality Control material for Teicoplanin. This is suitable for all Teicoplanin assays, and has been validated against the QMS Immunoassay on Roche Cobas platform.
It is a liquid stable, tri-level QC, packaged as 3 x 2ml with 12 months expiry from date of manufacture. Being a 3rd party QC, it helps labs fulfill this criteria in ISO 15189.