19 February 2021
An important indicator of ovarian reserve is a blood test called Anti Mullerian Hormone. It is a very sensitive marker of ovarian reserve used by fertility clinics, with the analysis performed in the Clinical Laboratory.
The ADP Tri-Level anti-Mullerian Hormone (MIS) Quality Control set is designed to monitor the performance of AMH immunoassays. This quality control has been validated against the Roche (ECLIA) immunoassay on the Roche Cobas analysers. This is a new product that enables clinicians to have a 3rd party QC for their laboratories.
The quality control has been developed in conjunction with the Royal Victoria Hospital in Belfast, Northern Ireland. The levels have been chosen to reflect critical clinical cut-off values around 12 pmol/L, a mid assay and an elevated level of 47 pmol/L, to check the linearity of the assay.
Stored frozen, defrosted it is stable for 7 days. The kit contains 2 bottles of each level, 3 levels, and 2 mls per vial ( 2 x 3 x 2ml).
Contact ADP Diagnostics for further information or for more information on our range of niche quality controls click here.
Further references:
References: Clinical Biochemistry Volume 49, Issue 3, February 2016, Pages 260–267
(The 2013 National Institute for Health and Care Excellence (NICE) clinical guideline for fertility and the European Society of HumanReproduction and Embryology (ESHRE) recommend AMH measurement as one method to predict ovarian response to gonadotropin stimulation [12] and [13]. The NICE guideline states that AMH concentration of ≤ 0.75 ng/ml (≤ 5.4 pmol/L) indicates a low ovarian response to stimulation, whereas AMH concentration ≥3.50 ng/ml (≥ 25.0 pmol/L) indicates a high response [12]. It follows that precise and accurate AMH measurement in these intervals is a prerequisite for reliable interpretation of AMH results in a clinical setting)

12 February 2021
PSA Ultra Low Quality Control is a product that we developed in response to numerous requests for a low level control to keep pace with the ever-increasing sensitivity of PSA assays. The control is widely used in Ireland by cancer testing centres as it is ideal for monitoring PSA levels post-prostatectomy.
The control is validated against the Roche immunoassay on the Roche Cobas® analyser.
The ‘PSA: Low level’ control is manufactured as stabilised serum from human origin. The concentrations of the QC have been chosen to ensure optimal assay performance at a critical sensitivity level.
A low level or undetectable PSA level is defined as ≤0.06 ng/mL or 0.02ng/mL by the ‘American Society for therapeutic radiology and oncology consensus panel’. As defined by the intuition it is agreed that a 6-12 month ‘undetectable PSA’ level is a prediction of disease-free survival ‘DFS’. Therefore an ultra-sensitive assay is beneficial.
Reference: Int. J. Radiation Oncology Biol. Phys Vol.37 No.5 pp 1035-1041, 1997
The control is stable up to the expiry date when stored at 2-8°c. When opened the control is stable for a period of 14 days when stored at 2-8°c. The kit size is 10 x 1ml.
Contact us for instrument specific values or further information.

03 February 2021
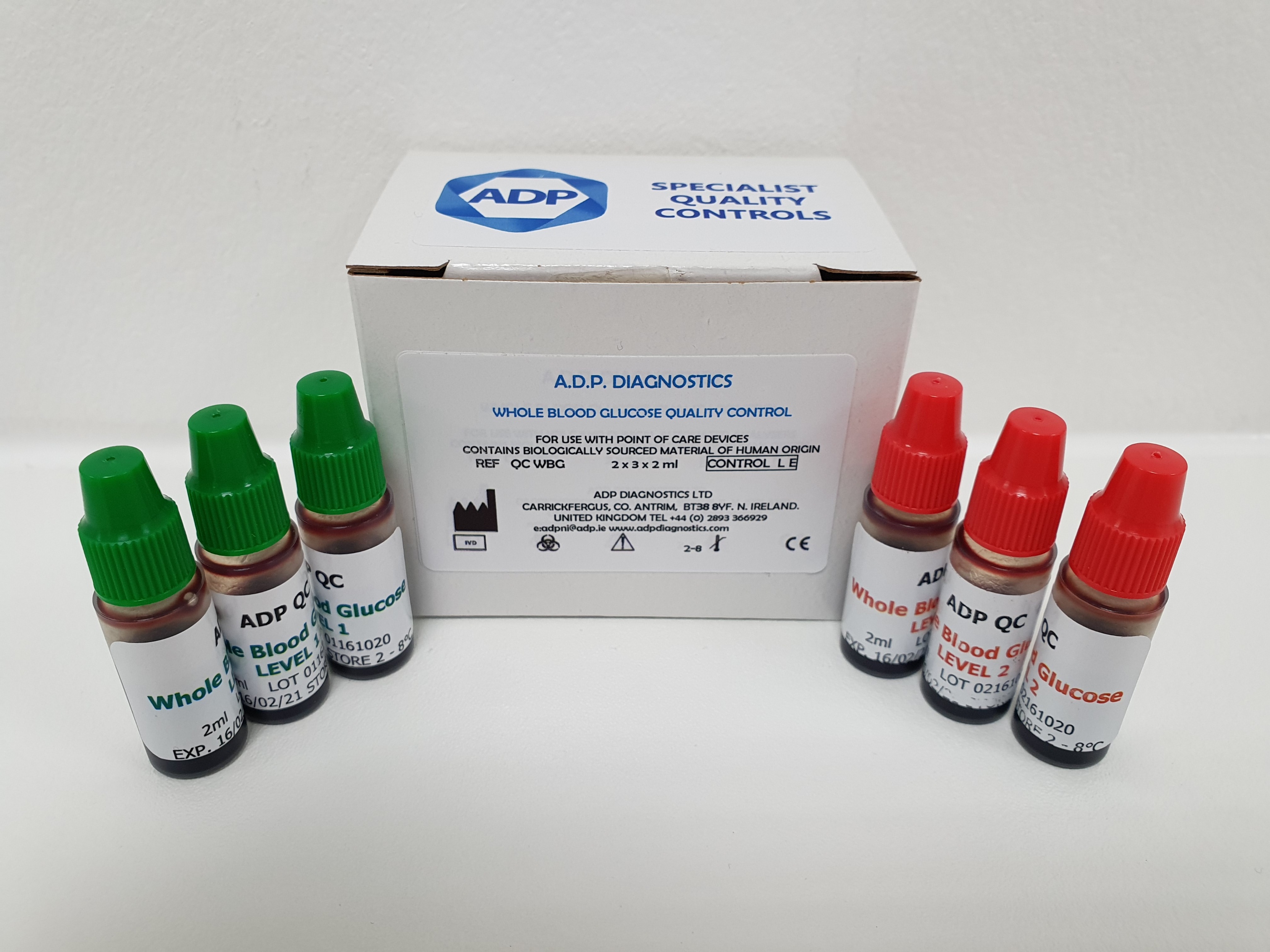
After months of investment, research, development and trials, ADP Diagnostics in has successfully launched its new whole blood glucose quality control, a world first. The impact of this new product is significant, in that for the first time it enables clinicians and point of care specialists to use a quality control material for POC glucose testing, that is as close as possible to a patient sample.
The whole blood glucose quality control is supplied as a 2-level control x 3 vials of each level, vials are 2ml. The control is compatible with all major manufacturers’ Glucometers and instrument specific values are available. Once opened, the vials are stable for 4 months. Vials should be stored at 2-8C.
The new whole blood glucose control is of interest to any laboratory that implements a policy of continuous improvement in clinical excellence. Using this control also enables the laboratory to comply with ISO15189 guidelines which stipulate:
“The laboratory shall use quality control materials that react to the examining system in a manner as close as possible to patient samples. Quality control materials shall be periodically examined with a frequency that is based on the stability of the procedure and the risk of harm to the patient from an erroneous result.”
(Reference: ISO 15189:2012(E): 5.6.2.2 Quality control materials)
ADP Diagnostics has built a strong reputation for innovation and clinical excellence, having already launched a number of world first quality control products including a single vial Serum Indices Quality Control, and a Xanthochromia Quality Control. The company’s products are being used throughout Ireland, UK, and several other international markets. ADP Diagnostics has become a key supplier of niche quality control sera to a number of National Quality Assessment Schemes worldwide. The control is currently being used by Labquality Finland, one of Europe’s major EQA scheme organisers.
Contact us for instrument specific values or for further information.
*The whole blood glucose quality control has been developed in association with Dr L. McGrath of Torax Biosciences.